- METRC continues to move new studies forward
METRC has sponsored 36 prospective multicenter studies to date. Of these, 12 are ongoing; 7 have completed data collection and analyses are in progress; and 17 are fully complete. The Consortium continues to seek funding to carry on its vital mission. Eight new grant proposals have been submitted this year and 2 additional proposals are in progress.
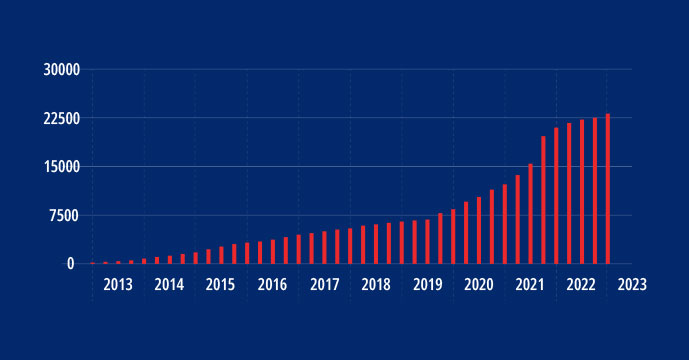
- Over 23,000 Patients
Since 2012, METRC has worked with more than 23,000 patients across 82 different sites and 38 studies.
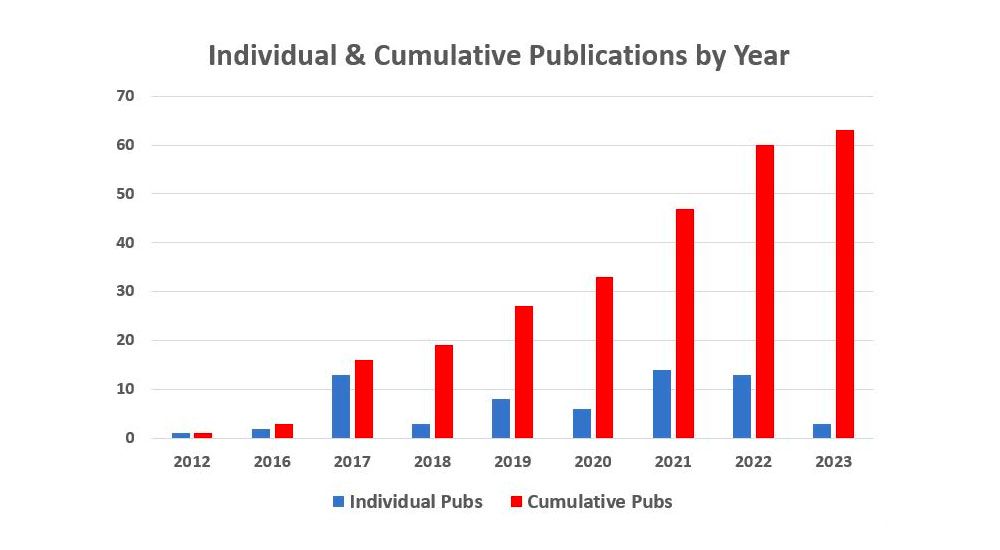
- Annual and Cumulative Publications
Our publication output continues to grow. This year METRC will present 13 podiums at the Orthopaedic Trauma Association Annual Research Meeting in Seattle. All of the podium presentations will be become METRC publications over the course of the next year.

Featured Achievement
METRC accepts The Clinical Research Forum’s Top 10 Clinical Research Achievement Award for the PREVENT CLOT study
In addition, PREVENT CLOT won a further honor, the Distinguished Clinical Research Achievement Awards
- Read more

Featured achievement
Kappa Delta Elizabeth Winston Lanier Award
Major Extremity Trauma Research Consortium Receives Prestigious Award for Advancing the Field of Clinical Trials in Trauma Orthopaedics.
- Read more
.png)
Upcoming Events
METRC had a Major Showing at 2023 OTA Meeting
For the 2023 Orthopaedic Trauma Association Annual Meeting in Seattle, METRC presented 22 abstracts.
- Read more